 
La deleción heterocigota de los genes HIRA, GNB1L o TUBA8 podría desempeñar un papel importante en el desarrollo del tabique ventricular. La determinación del genotipo de microsatélites identificó la LOH de HSA22q11 en varios tipos de CPC. 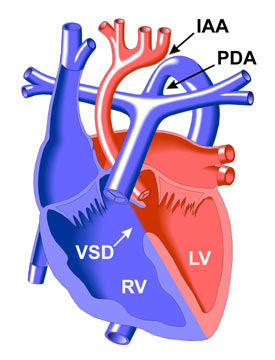
El análisis de dosis realizado para siete genes candidatos indicó una deleción heterocigota recurrente de los genes HIRA, GNB1L y TUBA8 en 16 pacientes con CIV que presentaban LOH de HSA22q11. Se observaron LOH de HSA22q11 en 17 de 29 pacientes con CIV y en 3 de 4 familias con ambos hijos afectados por CPC. En 42 (49,4%) de los 85 pacientes con CPC se identificó la presencia de al menos una LOH en la región HSA22q11. Se aplicaron reacciones en cadena de polimerasa cuantitativas para el análisis de dosis a siete genes candidatos de 16 pacientes con LOH de HSA22q11. 
Las estrategias de bioinformática permitieron caracterizar más detalladamente siete genes candidatos situados en esa región. Se efectuó un examen de detección de la LOH en la región HSA22q11 mediante determinación del genotipo de microsatélites (n = 10). Se investigó a un total de 82 familias con CPC, en las que había 261 individuos (85 pacientes, 176 hermanos y progenitores). Con objeto de identificar los genes candidatos situados en el HSA22q11 asociados a la CIV, se analizó la pérdida de heterocigosis (LOH) mediante determinación del genotipo de microsatélites y análisis de dosis de siete genes candidatos. La comunicación interventricular (CIV) es una de las principales formas de cardiopatía congénita (CPC) en los individuos con el síndrome de deleción del cromosoma 22q11 del Homo sapiens (HSA22q11). Since CHD can be a familial disease, screening the siblings of a proband for LOH of HSA22q could be valuable for early diagnosis and treatment. Heterozygous deletion of HIRA, GNB1L or TUBA8 genes might play an important role in ventricular septum development. Microsatellite genotyping identified LOH of HSA22q11 in several types of CHD. Dosage analysis of the seven candidate genes showed recurrent heterozygous deletion of HIRA, GNB1L and TUBA8 genes in 16 VSD patients with a LOH of HSA22q11. Moreover, LOH of HSA22q11 was found in 17 out of 29 patients with a VSD and in three out of four families with two offspring affected by CHD. Overall, 42 out of 85 patients (49.4%) with CHD had at least one LOH in the HSA22q11 region. Quantitative polymerase chain reaction analysis was used to determine the dosages of the seven candidate genes in 16 patients with LOH of HSA22q11. Bioinformatic strategies were used to characterize seven candidate genes located within this region in greater detail. All were screened for LOH in the HSA22q11 region by microsatellite (n=10) genotyping. The study involved 82 families with CHD, which included 261 individuals (85 patients and 176 siblings and parents). The objective was to identify candidate genes associated with VSD located within HSA22q11 by analyzing loss of heterozygosity (LOH) using microsatellite genotyping and by gene dosage analysis in seven candidate genes. Ventricular septal defect (VSD) is one of the major forms of congenital heart disease (CHD) in individuals with Homo sapiens chromosome 22q11 (HSA22q11) deletion syndrome. However, in this case the Ventricular Septal Defect (VSD) allows mixing of oxygen-rich blood from the left ventricle into the right ventricle, which pumps blood into the pulmonary artery.Introduction and objectives. The pulmonary artery normally carries oxygen-poor blood to the lungs, so it might seem that blood entering the lower aorta from this vessel (through the PDA) would not carry enough oxygen to the lower body. (The Patent Ductus Arteriosus is a feature of the circulatory system that usually closes soon after birth.) However, some blood does enter the lower part of the aorta because of a small vessel, known as the Patent Ductus Arteriosus (PDA), that connects the lower part of the aorta with the pulmonary artery. Second, there is a hole, called a Ventricular Septal Defect (VSD in diagram), in the muscle wall (septum) that separates the two ventricles, or pumping chambers of the heart.īecause the aorta is interrupted and cannot carry blood from the left ventricle to the lower body as in a normal heart, it might seem that the child with this anomaly could not survive. The aortic arch is the part of the aorta (the major vessel that carries oxygen-rich blood from the heart to the body's tissues) that curves directly above the heart and begins the descent to the lower body. First, the Aortic Arch does not form a complete tube and is divided, or "interrupted" (see upper arrow in diagram). This rare genetic disorder involves two defects. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
